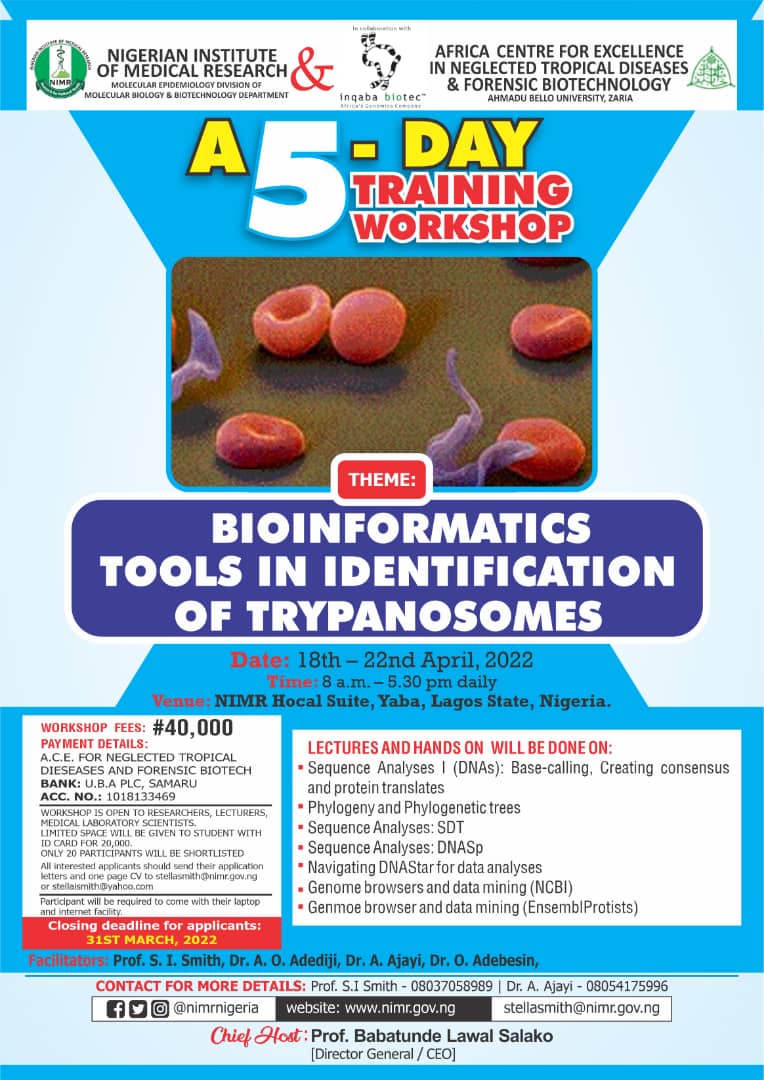
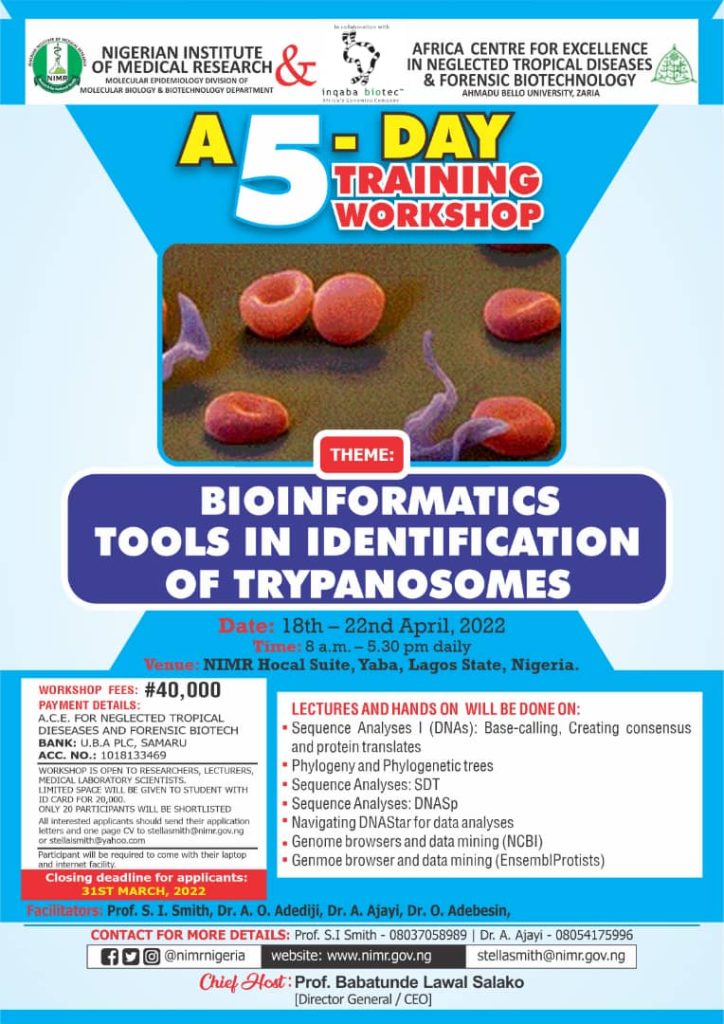
Theme: The Impact of Science in the Covid-19 Era
Guest Speaker: Prof. Babatunde Lawal Salako – Director General, Nigerian Institute of Medical Research, Yaba;
Chief Host: Prof. Jeremiah Oludele Ojediran – Vice-Chancellor, Bells University of Technology;
Host: Dr. Augustine Omoike – Ag. Dean, COLNAS
- Date & Time:
- April 6, 2022
- 1:00 pm
- Venue: BUPF International Conference Centre